
Elderly Launceston participants in a world-first medical trial to determine whether aspirin improves quality of life were given a progress update on Friday.
Subscribe now for unlimited access.
$0/
(min cost $0)
or signup to continue reading
The Aspirin in Reducing Events in the Elderly project, led by Monash University in Melbourne, started in 2010 and involves more than 2000 Tasmanians.
The study is investigating if taking low-dose aspirin daily can preserve good health through fighting common disease including cardiovascular disease, dementia, depression and some cancers.
It examines whether the benefits of the practice outweigh associated risks including bleeding.
The trial’s participants take 100mg of aspirin or a placebo tablet daily and they undergo annual study health checks for an average of five years.
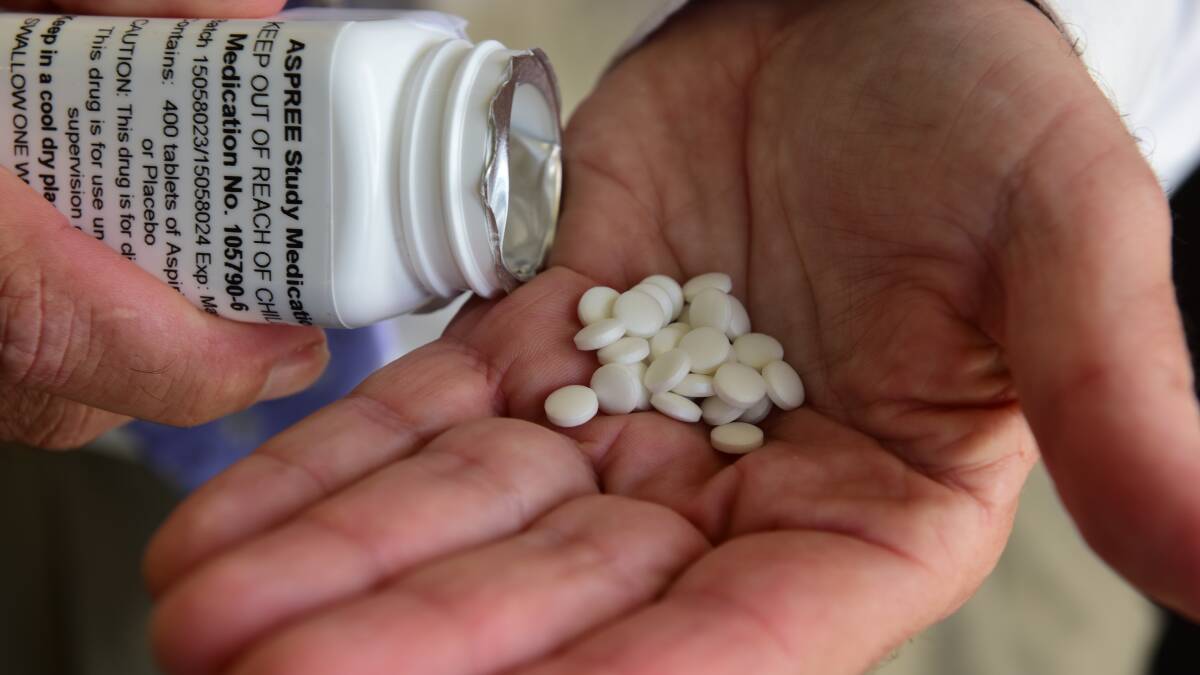
Trial researchers, participants and their GPs do not find out who is taking which until the trial’s conclusion.
All participants in the trial are aged over 70.
The University of Tasmania’s Menzies Institute for Medical Research is a collaborating centre with the trial.
Friday’s update was provided in a presentation by ASPREE chief investigator in Tasmania, Professor Mark Nelson, who said the first person recruited to the trial in 2010 was Tasmanian.
“More Tasmanians are participating per head of population than anywhere else in the world, this [state] has got the greatest volunteerism of anywhere in the world, so well done Tasmania,” he said.

He said he believed community spirit underpinned participation in the “public good” study. The study’s results would be “entirely influential”, he said.
Professor Nelson provided updates on the study and gauged community feedback at the function.
“It’s a community study ... so two-way community feedback is [vital],” he said.
He told attendees about the progress of 15 ASPREE sub-studies, which look at the effect of aspirin on conditions including hearing loss.
More than 550 residents of the Launceston region enrolled in ASPREE, supported by their GPs.
The trial has 16,703 participants throughout Australia’s south-east and 2411 participants from the US.
The trial is due to end in December 2017 and early results are expected mid-2018.
More than 200 local trial participants and guests attended the event for an update at the Tamar Function Centre in Riverside.


