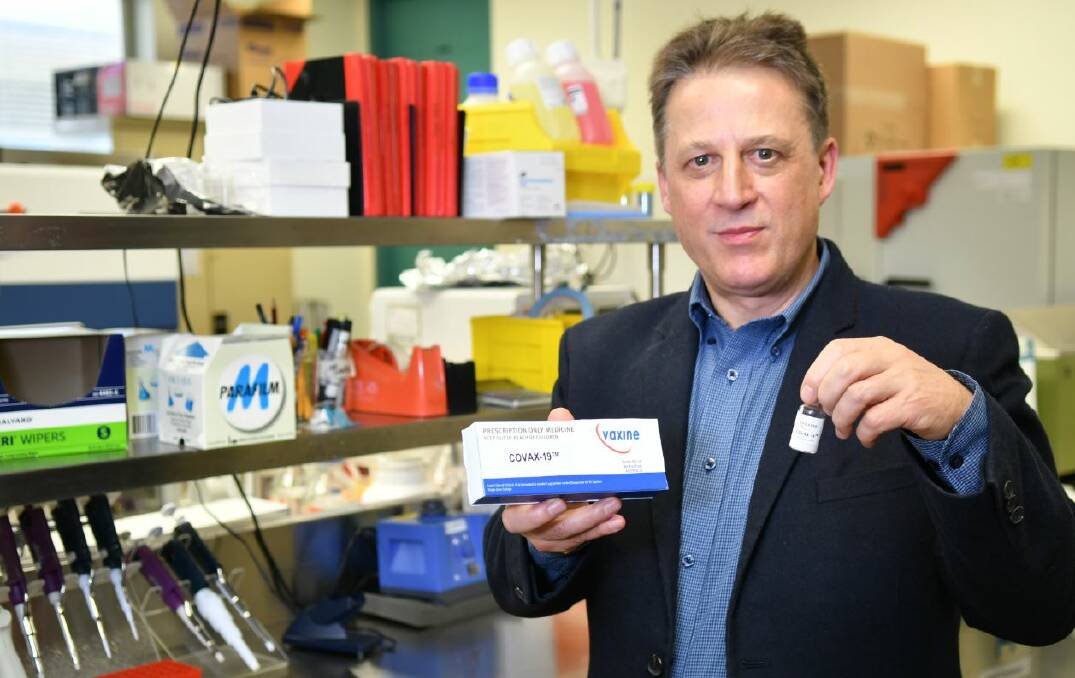
A Launceston-born endocrinologist has developed the first Australian vaccine capable of protecting against the Delta strain of the novel coronavirus
Subscribe now for unlimited access.
$0/
(min cost $0)
or signup to continue reading
In July 2020, University of Tasmanian Alumni, and Flinders Medical Centre director of endocrinology Nikolai Petrovsky developed COVAX-19 - the first COVID-19 vaccine developed in Australia to enter human trials.
The vaccine is based on a protein produced in insect cells was developed by Vaxine, a research company founded by Professor Petrovsky 18 years ago.
He said the development of the synthetic version of the COVID-19 spike protein was the culmination of 20 years of researching and developing vaccines from around the world.

Funded by the United States government through the National Institutes of Health, Professor Petrovsky explained Vaxine has been developing vaccines for some of the most significant public health emergencies since the early 2000s.
"In 2004 there were the anthrax letter attacks in the United States," he said.
"There was a recognition that more work and research into producing vaccines that could protect against terrorist attacks with biological weapons needed to be done, but also vaccines around pandemics because around that time we had the SARS coronavirus outbreak, '' he said.
"We did eventually produce successful anthrax and SARS vaccines, and I've been working on pandemic vaccines ever since."
Professor Petrovsky said he successfully developed vaccines for other pandemics, including avian influenza, which included successful clinical trials in humans, and developed a vaccine for the MERS virus.
With an expert understanding of how viruses spread and evolve, Professor Petrovsky recognised in the early days of the coronavirus pandemic the potential health implications for the global community, and set about developing a vaccine.
"In January last year, I recognised that this was likely to be a major issue and a potential pandemic so we started developing a vaccine straight away," he said.
"We were one of the first in the world to be on top of the virus and have a vaccine in development as early as January-February last year.
"We started the first human trials here in Adelaide, it was the first actual vaccine developed in the southern hemisphere to go into humans, which was a big achievement. So not just the first in Australia, first in the southern hemisphere."
After successful phase one trials in Australia were completed, Vaxine partnered with Iranian pharmaceutical company CinnaGen to undertake clinical phase two and three trials in Iran, which was chosen for the trials due to the high number of cases the county was recording.
"They were running at about 1000 deaths a day and up to 200,000 cases per day. We're talking about one of the worst outbreaks anywhere in the world," he said.
"So obviously a terrible situation in Iran from a public health perspective, but actually the best place to go to test out an effective vaccine"
READ MORE: Northern Tasmanian sweeps tourism awards
Following successful clinical trials in Iran which Professor Petrovsky said recorded no adverse side-effects or deaths, COVAX-19 was approved for use in Iran.
While not approved for use in Australia Professor Petrovsky said Vaxine is in talks with the Therapeutic Goods Administration in the hopes of having the drug approved for domestic use in Australia, and has vaccinated himself against the coronavirus with his vaccine.

Our journalists work hard to provide local, up-to-date news to the community. This is how you can continue to access our trusted content:
- Bookmark www.examiner.com.au
- Make sure you are signed up for our breaking and regular headlines newsletters
- Follow us on Twitter: @examineronline
- Follow us on Instagram: @examineronline
- Follow us on Google News: The Examiner


